We often see solar panels on the roofs of houses, on street lights, on calculators, on watches and even on satellite satellites in space. We know that these solar panels generate energy with the help of sunlight and we use this energy for various purposes. In fact, the sun is a huge source of energy. It has been continuously transmitting energy for billions of years. It is a giant nuclear power plant that emits about 1400 watts of energy per square meter of the earth's surface. But, do you know how these solar panels convert sunlight into electricity? Or how do these solar panels work as energy? Let's learn more about it today.

Firstly, the question is what is a solar cell? A solar cell is basically an electronic device that converts light energy directly into electricity through a photovoltaic effect. These solar cells are also called photovoltaic cells and this system is called photovoltaic or PV system. A package made up of many solar cells is called a solar panel. One solar cell can produce about 0.5 volts of electricity and where each cell is connected to others in modules. Solar panel voltage can be increased by increasing the number of solar cells in it. In order to get enough volts, many solar panels are arranged together in an array.
The most efficient solar cell yet can only convert 46% of the available sunlight to the electricity. And the ones that are used in all the commercial cases are usually maximum 15-20% effective. Basically, these solar panels work like a battery. However, the main difference with batteries is that battery cells usually generate electricity from the chemical reactions inside them and solar panels generate electricity directly from light through the PV system. Now, let's find out how these solar cells work to generate electricity.
Solar cells are mainly made of semiconductor material. Generally, crystalline silicon is used here as a semiconductor material. We know that silicon is the second highest abundant element in the world. As a solar cell, this crystalline silicon is embedded in two conductive layers. Each silicon atom is bound by 4 strong bonds to the silicon next to it. And, these bonds basically help the electrons to move in the right place.
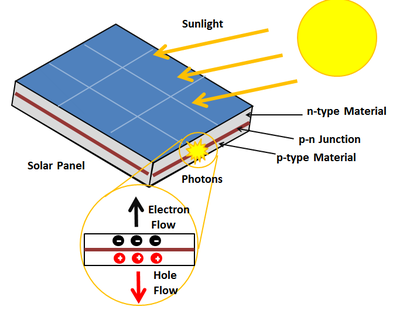
These silicon solar cells usually use two types of layers. One is N-type and the other is P-type. N-type silicon is relatively thinner and has a high electron concentration. And, P-type silicon has extra space for electrons which are called Holes. The P-N junction is formed by combining these two types of silicon layers. So that the electrons can move around there easily. This generates a negative charge on one side and a positive charge on the other side. This is the real thing that is caused by sunlight. As we know, there are many small particles flowing in the sunlight. These are called photons. When one of these photons strikes a cell of silicon with great force, the electrons in the bond of silicon move away leaving a hole. As a result, those negatively charged electrons and positively charged holes are released and can move wherever they want.
An interesting thing that happens here is that due to the presence of electric fields at the P-N junction, they are transmitted only in one direction. The electrons move towards the N layer where the holes move towards the P-layer. In this way, many clusters of electrons are controlled and transmitted together by a thin metal finger on the top of the solar panel, which is made of aluminum. This is how those electrons flow through an external circuit and participate in the use of electricity.
The funding to build the infrastructure and a good deal of space for solar energy technology can help the world have a better place of electricity with a much cheaper and better way.
Thanks for reading out !
Warm Regards,
Shahin Alam
COO at Utopia Educators


